It is incompatible with strong oxidizing agents and strong acids and bases. It can cause gastritis and ulceration when taken for a long time. Health effects / safety hazards: Aspirin is stable at room temperature, however it should keep dry to avoid its hydrolysis. The production of other medicines or chemical compounds such as 4-hydroxycoumarin. Aspirin is also an intermediate and raw material in Uses includes the rheumatic fever and Kawasaki disease. It is used mainly as anti-inflammatoryĪnd anti-pyretic but in the last decades it has been a very popular in cardiovascular diseases treatment. Uses: Acetylsalicylic acid is one of the most sold medicines around the world, since in 1897 Bayer laboratory gave the name of Aspirin and started its commercialization. Production (prostaglandins are molecules involved in inflammation process). Of action is due the inhibition of enzyme cyclooxygenase, causing the suppression of Prostaglandin It is soluble in water, ethanol, ethyl ether andĬhemical properties: Aetylsalicyl acid is known by its capacity as anti-inflammatory drug. Point is 135 ✬ and in higher temperature it decomposes.

Hydrolysis yielding salicylic and acetic acid. Physical properties: Acetylsalicylic acid is a colorless to white crystalline solid with a vinegar odor due its The hydroxyl group in the salicylic acid is replaced by and ester group. Preparation: Aspirin is synthesized through the esterification of salicylic acid by the acetic anhydride, thus It was synthesized by the first time in 1853 by the french So because salicylic acid produces the smallest amount of aspirin, salicylic acid is the limiting reactant.
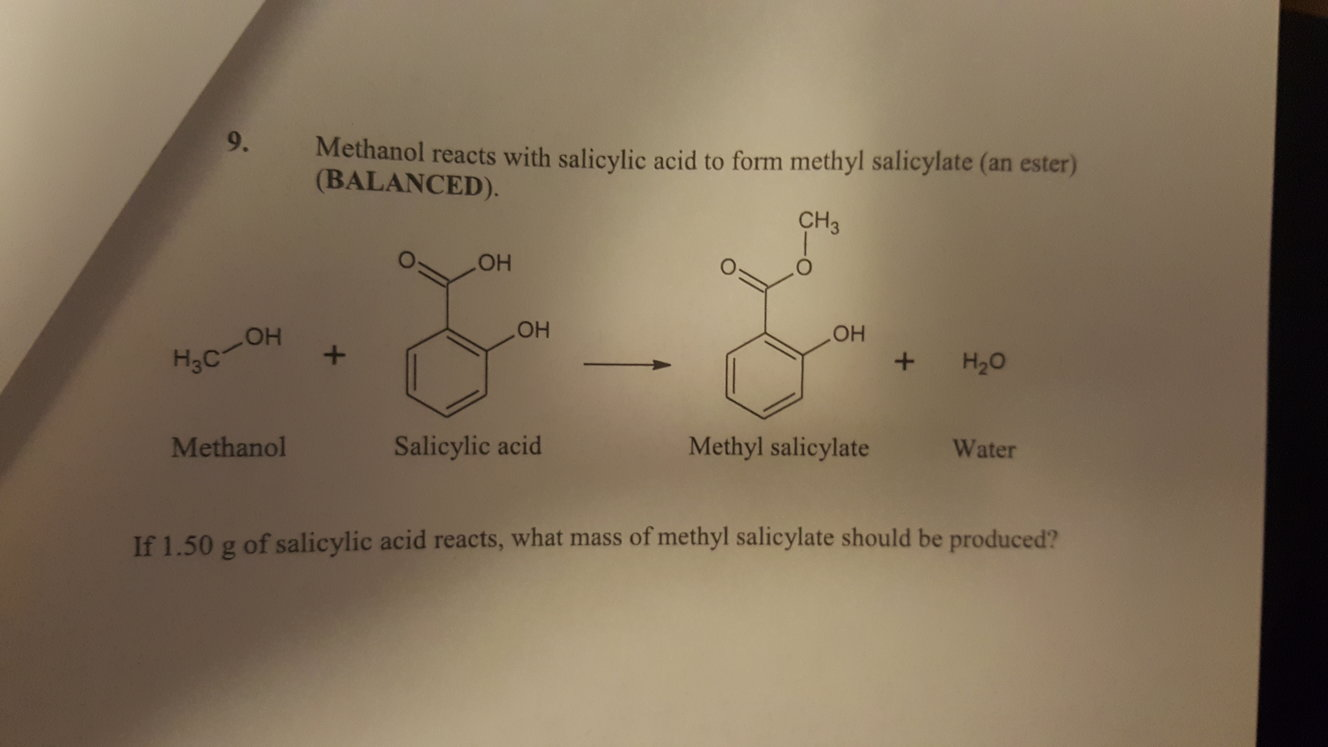
Ah, 180.16 g, and we get 1.63 g of aspirin. Occurrence: Aspirin can not be found in nature. Two moles of aspirin using the store Akiyama tree one toe, one, then moles of aspirin, 2 g of aspirin by multiplying by the molar mass of aspirin.

Its chemical structure can be written as below, in the common representations used for organic molecules. The molecular geometry is planar because the phenyl ring and the carboxylic groups which haveĪ sp2 hybridization. The molecule is formed by an aromatic ring which has two functional groups in position -orto: the first substituent is a carboxylic acid and the second an ester group. Formula and structure: The acetylsalicylic acid chemical formula is C 9H 8O4 and the extended formula isĬH 3COOC 6H 4COOH. Determine the mol of salicylic acid used if the molar mass of aspirin is 180.16 g/mol and the molar mass of salicylic acid is 138.12 g/mol.
